Seminar: Kim, Tae Gyun Ph. D.
Date
Location
Description
Structural Investigation on Human Mitochondrial Calcium Channels and QPRT Enzyme as Brain Detoxifier
Ph.D. Kim, Tae Gyun
Steitz Center for Structural Biology
Gwangju Institute of Science and Technology
November 16, 2015
Abstract
Part I
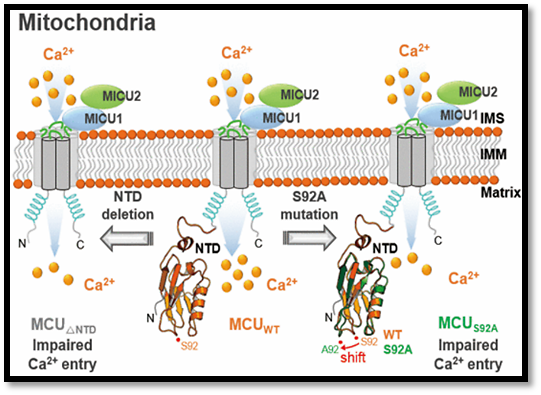
The mitochondrial calcium uniporter (MCU) is responsible for mitochondrial calcium uptake and homeostasis. It is also a target for the regulation of cellular anti-/pro-apoptosis and necrosis by several oncogenes and tumour suppressors. I introduce the high-resolution crystal structure of the MCU N-terminal domain as a novel fold. The assembly of the mitochondrial calcium uniporter complex and the interaction with the MCU regulators such as the mitochondrial calcium uptake-1 and mitochondrial calcium uptake-2 proteins (MICU1 and MICU2) are not affected by the deletion of MCU NTD. In addition, I introduce structural and biochemical characterization of trimeric EF-hand motif of leucine-zipper EF-hand containing transmembrane protein 1 in mitochondrial inner membrane region. Even though these channels are little complementary to mitochondrial calcium influx, structural and functional studies of both channels can further explore the mechanistic approaches on mitochondrial calcium ion channels.
Part II
Quinolinate phosphoribosyltransferase (QPRT) catalyzes the production of nicotinic acid mononucleotide, a precursor of de novo biosynthesis of the ubiquitous coenzyme nicotinamide adenine dinucleotide. QPRT is also essential to maintain homeostasis of quinolinic acid in the brain, a possible neurotoxin causing various neurodegenerative diseases. Despite extensive studies of QPRT have been performed, molecular basis of the reaction catalyzed by human QPRT is still mostly unknown. Here we present the crystal structures of hexameric human QPRT in apo form and its complexes with reactant or product and revealed that the interaction between dimeric subunits is dramatically altered during the reaction process by conformational changes of two flexible loops in the active site at the dimer-dimer interface. In addition, the N-terminal short helix α1 was identified as a critical hexamer stabilizer. The structural feature, size distribution analysis and heat aggregation property of the full-length enzyme and the enzyme lacking helix α1 strongly suggest that human QPRT acts as a hexamer for cooperative reactant binding via three dimeric subunits and maintaining stability. By comparing human QPRT structures in the apo and complex forms, we propose a drug design strategy targeting malignant glioma.
Attachments
Subscribe to the OIST Calendar: Right-click to download, then open in your calendar application.



